|
We successfully provide students with intensive courses by India's top faculties and personal mentors. Physics Wallah also caters to over 3.5 million registered students and over 78 lakh+ Youtube subscribers with 4.8 rating on its app. We also provide extensive NCERT solutions, sample papers, NEET, JEE Mains, BITSAT previous year papers, which makes us a one-stop solution for all resources. Physics Wallah is India's top online ed-tech platform that provides affordable and comprehensive learning experience to students of classes 6 to 12 and those preparing for JEE and NEET exams. Ecell is always positive in a galvanic cell because the anode always has a negative E and the cathode always has a positive E. These devices can be used to obtain the chlorine gas and the metallic sodium from the aqueous solutions of the sodium chloride (common salt).Īns. Electrolytic cells can be used to produce the oxygen gas and the hydrogen gas from the water by subjecting it to an electrolysis. What are the uses of electrolytic cells?Īns. Therefore, the negatively charged ions flow towards the anode, that is positively charged. This results in the deposition of an positively charged ions into the cathode. When the external electric current flows into the cathode of an electrolytic cell, the resulting negative charge will attracts the dissociated positive ions present in an electrolyte. E cell is negative for electrolytic cell.Īns. Is E cell positive in electrolytic cell?Īns. It may be noted that the industrial production of high-purity copper, high-purity zinc, and high-purity aluminum is almost always done through electrolytic cells. Such electrochemical cells are also used in electrolytic recovery processes. They are also used to extract aluminum from bauxite.Īnother application of electrolytic cells is in electroplating, which is the process of forming a thin protective layer of a specific metal on the surface of another metal.Įlectrorefining of many non-ferrous metals is carried out using electrolytic cells. The primary use of electrolytic cells is for producing oxygen gas and hydrogen gas from water. Thus, NaCl can be subjected to the electrolysis in an electrolytic cell to generate the metallic sodium and chlorine gas as the products. The associated chemical equations and overall cellular reaction are given below. This results in the formation of the chlorine gas (Cl 2) at the anode (that is accompanied by the release of 2 electrons, thus completing the circuit). This results in the formation of sodium metal at the cathode.Īt the same time, a chlorine atoms are attracted towards the positively charged cathode. The positively charged sodium cations are attracted toward the negatively charged cathode. When an electric current passes into the circuit, the cathode becomes rich in a electrons and then develops the negative charge. Here, two inert electrodes are immersed in the molten sodium chloride (that contains dissociated Na + cations and Cl – anions). 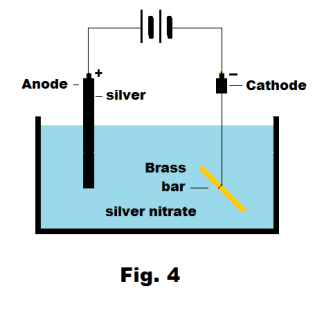
Molten sodium chloride (NaCl) can be applied to the electrolysis with the help of an electrolytic cell, as mentioned below. Working and Diagram of an Electrolytic Cell It contains positively charged anode and negatively charged cathode It contains negatively charged anodes and positively charged cathodes It converts electrical energy into chemical energy It converts chemical energy into an electrical energy
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
